 
In this blog post we discussed some basics of quantum mechanics through the hydrogen atom and saw how to reproduce results using the Schrödinger Equation interface in COMSOL Multiphysics ®. 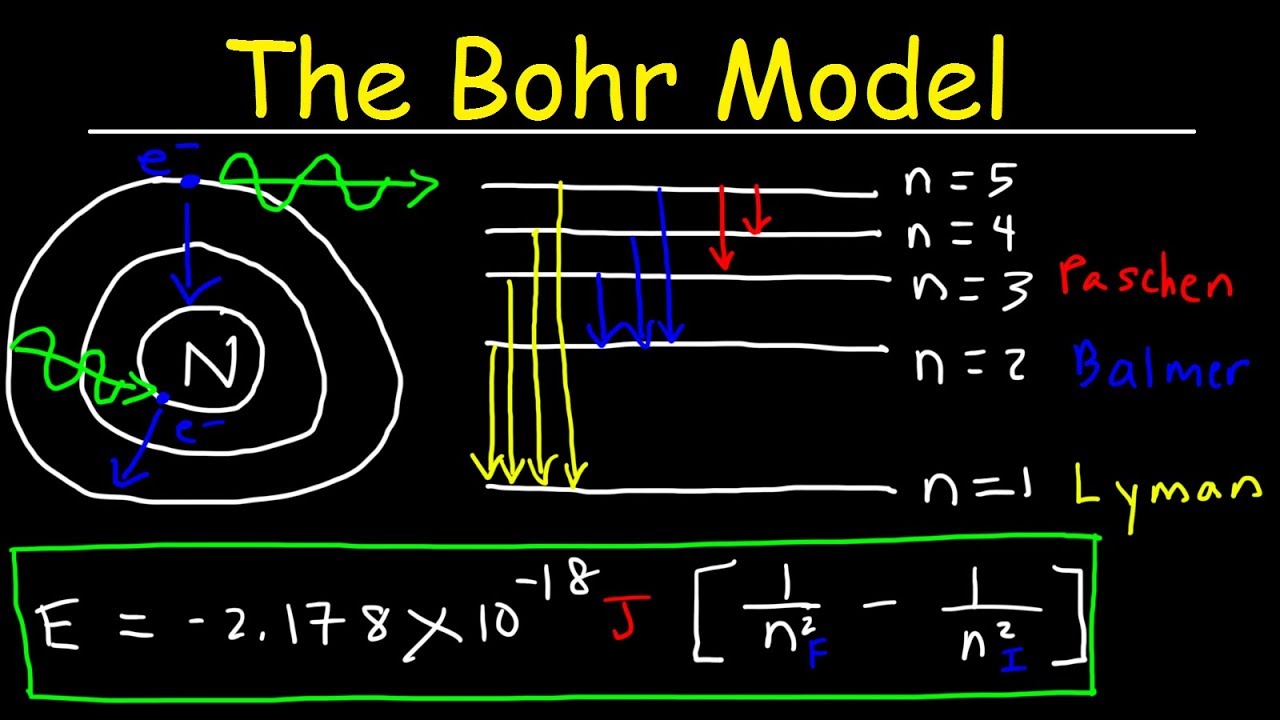
Orbital shapes in the presence of an external electric field along the z -axis. Unlike multielectron atoms, the hydrogen atom with its single electron is, to an excellent approximation, described by the time-independent Schrödinger equation (TISE): 
Since then, quantum mechanics has become the most verified theory in physics. One of the early demonstrations of the power of quantum mechanics was the experimental confirmation of the results for hydrogen (see below) by spectroscopic measurements. The COMSOL Multiphysics ® user interface with the Graphics window showing the hydrogen atom model, discussed further in the following section. This is mathematically analogous to a standing wave: the wave must be single-valued, which is possible for only certain fixed wavelengths, i.e., energies. Quantum mechanics resolves the conundrum by establishing that bound electrons can only possess certain fixed energy values, prohibiting a gradual loss of energy by radiation. A charged particle in accelerating motion (such as an electron orbiting a nucleus) should produce electromagnetic radiation, thus gradually losing energy and resulting in the inevitable collapse of the electron into the nucleus. The existence and stability of atoms, which consist of a positively charged nucleus surrounded by a cloud of negatively charged electrons, cannot be explained with classical physics. In this blog post, we take a look at perhaps the most significant exact results of quantum mechanics - the ground state and the first few excited states of the hydrogen atom - and go over how to reproduce these results using the COMSOL Multiphysics ® software. It may be fair to say that no other theory has shaken our understanding of reality quite as profoundly. As a result, mvr = nh/2π, where m = electron mass, v = electron tangential velocity, and r = radius of Bohr energy levels.Īnother example of quantization is atomic line spectra.Quantum mechanics ushered in the era of modern physics, specifically during the first decades of the 20 th century. This integral multiple is known as the hydrogen atom’s primary quantum energy level. An electron’s angular momentum in an orbit is an integral multiple of h/2π.When an electron may jump from one orbit to another with more energy absorption but from one orbit to another with lower energy emission.An electron remains in a specific orbit in which no energy is emitted or absorbed.An atom, such as a hydrogen atom, has numerous stable circular orbitals in which an electron can remain.ISRO CS Syllabus for Scientist/Engineer Exam.ISRO CS Original Papers and Official Keys.GATE CS Original Papers and Official Keys.DevOps Engineering - Planning to Production.Python Backend Development with Django(Live).Android App Development with Kotlin(Live).Full Stack Development with React & Node JS(Live).Java Programming - Beginner to Advanced.Data Structure & Algorithm-Self Paced(C++/JAVA).Data Structures & Algorithms in JavaScript.Data Structure & Algorithm Classes (Live). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
